Eli Lilly has updated the design of the Mounjaro KwikPen. Your medication is not changing; only the pen's internal mechanism. Here is everything you need to know.
On 6 March 2026, Eli Lilly, the manufacturer of Mounjaro, announced a physical update to the KwikPen device used for your weekly weight loss injections.
This change relates entirely to the pen hardware. The weight loss medication inside, including the active ingredient tirzepatide, is completely unchanged. Your dose strength and concentration remain exactly the same.
Modified pens will begin arriving in the UK from April 2026. This article explains exactly what you will see when you open your next box, and what it means for your treatment.
What has changed with the new Mounjaro pen?
The main change is internal. Eli Lilly has redesigned the pen's plunger mechanism to reduce the amount of liquid left over after your fourth and final dose. This makes it significantly clearer when the pen is empty and ready for safe disposal.
The updated pen will replace the previous version gradually over the coming months. There are three specific visual changes you may notice.
These are intentional design improvements from the manufacturer and they do not mean your pen is broken or faulty.
The 3 design changes
- Plunger position: The plunger now starts further down the cartridge, which can make it look as though there is less liquid inside. There is not. The pen still contains your full four doses.
- Extended plunger: A new internal extension piece sits behind the plunger, making the mechanism appear longer than in previous models.
- End-of-pen indicator: After your fourth dose, the plunger now reaches the bottom of the cartridge, giving you a clear visual confirmation that the pen is empty and ready to dispose of.
See below a diagram showing the initial pen design alongside the updated version:

How does the additional component affect the plunger position in an unused pen and after each dose?
The plunger in the modified Mounjaro KwikPen appears further down in the cartridge than in the initial device. Following the completion of the 4 fixed 0.6mL doses, the plunger will appear near the bottom of the cartridge, and the amount of leftover medicine will be minimal. This is a normal and the intended outcome of the modified design.
See below diagrams showing KwikPen plunger positions in the initial pen design vs the updated version:
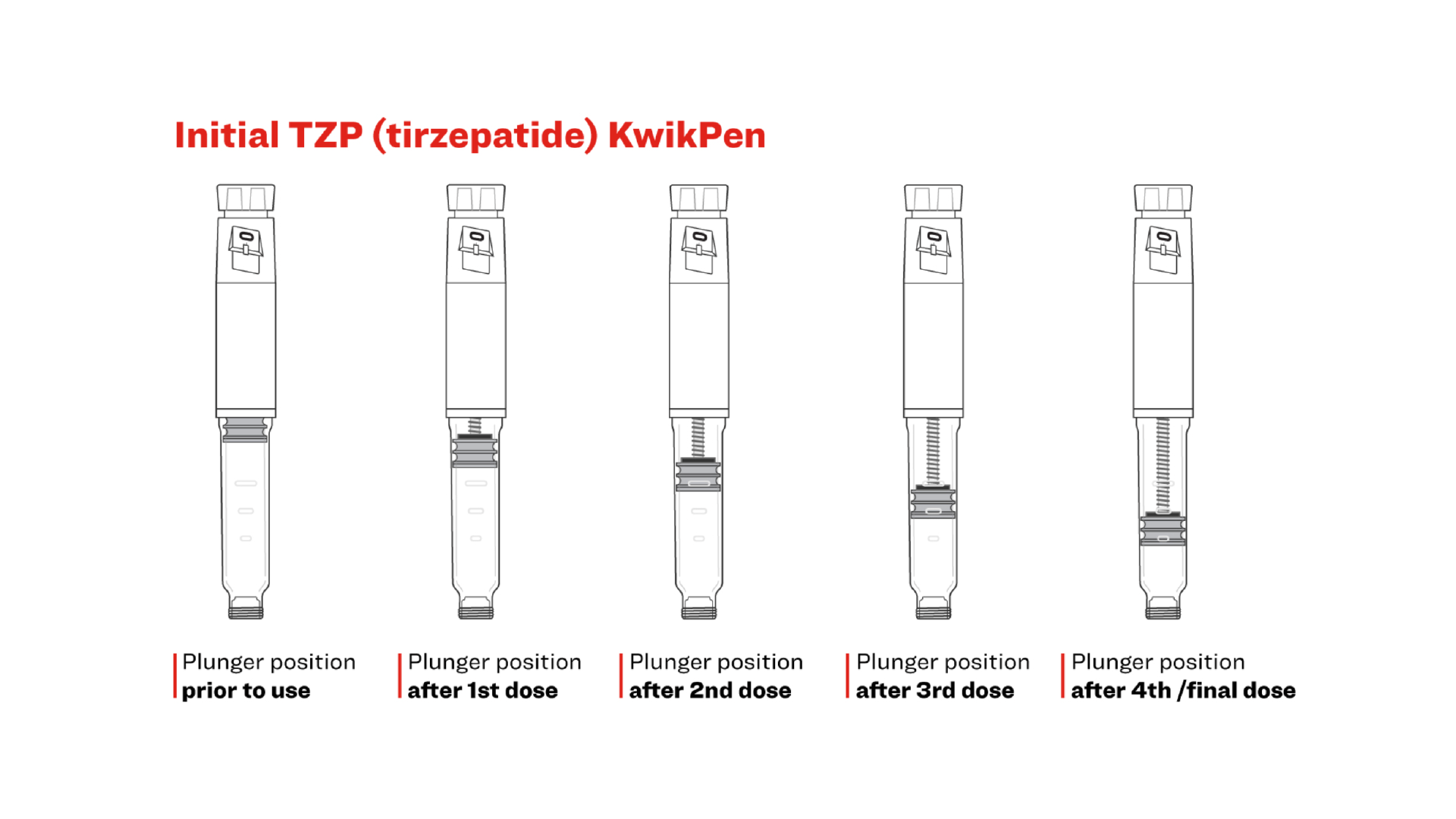

* These illustrations depict the approximate plunger positions
* Do not use the lines on the cartridge as does indicators
Following completion of 4 fixed doses
See below diagram showing plunger position after delivery of the 4th dose

What has stayed the same?
Your medication is exactly the same as before. Tirzepatide, the active ingredient in Mounjaro, remains identical in concentration and formulation. Your prescribed dose strength is unchanged, whether that is 2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg or 15 mg.
Only the internal plunger mechanism has changed. How you use your pen, including priming steps, dose dialling, and injection method, remains exactly as instructed by your Morrisons Clinic prescriber.
You can continue your usual weekly injection routine with confidence.
What about the ‘golden dose’ or ‘fifth dose’?
You may have heard of a “golden dose” or “fifth dose”: terms that refer to leftover liquid in the original KwikPen design that some patients attempted to use as an additional injection.
Using the golden dose is not recommended, and never has been. It goes against the medications licensed dosing requirements. The residual liquid in the old pen was an unmeasured buffer designed for device priming, not treatment. There is no guarantee it would provide an accurate or effective dose.
Attempting to access leftover liquid also carries a meaningful risk of bacterial contamination. Doing so requires tampering with the pen in a way that is not intended by the manufacturer.
The new plunger design removes this ambiguity entirely. After your fourth dose, only a negligible amount of liquid remains, not enough to constitute a usable dose. This brings your pen in line with the version already used in markets including the USA and Germany.
Why has Eli Lilly made this change?
The primary reason is clarity. In the previous pen design, residual liquid sometimes caused patients to question whether their device had worked correctly, or to wonder whether they should attempt a fifth dose.
By minimising leftover liquid, the pen’s “empty” state is now immediately obvious. When the plunger reaches the bottom of the cartridge, treatment for that pen is complete. No guesswork required.
This change brings the UK device in line with international standards and ensures patients receive exactly four accurate, consistent doses per pen, every time.
What does this mean for you in practice?
Because your dose, strength and injection routine are all unchanged, the transition should be entirely straightforward.
The first thing you are likely to notice is that the plunger looks “longer” or more “pushed in” than you are used to. This is normal. Both the old and new pen versions are safe and work correctly for your treatment. If you receive either version during the transition period, there is nothing to be concerned about.
A few practical reminders
- No new prescription is required. Your existing Morrisons Clinic prescription covers the medication regardless of pen design.
- Keep your usual injection day and preferred injection sites.
- Check the Instructions for Use (IFU) leaflet in your new box. The diagrams will match the pen you are holding, making it easy to identify which version you have.
- If you previously used a golden dose or fifth dose, be aware that the new pen will not support this. Make sure your next month’s supply is ready immediately after your fourth dose to avoid any gap in treatment.
Is it safe to use any liquid left in the pen after four doses?
No. Official guidance remains that you should only use the four prescribed doses from your Mounjaro KwikPen. The device is designed and validated to deliver four 0.6 ml injections. Any liquid remaining after that point is residual waste and should not be used.
Eli Lilly cannot guarantee the accuracy of any additional dose beyond the four intended, and taking the pen apart to access residual fluid carries a risk of contamination. The modified design makes it much easier to follow official usage guidance by removing any temptation to save or extract leftover liquid.
Stick to the four-dose schedule provided by your Morrisons Clinic prescriber. This is the safest and most effective way to use your medication.
Have questions? We’re here.
If you receive a new pen and feel unsure about how it looks, or if anything about your Mounjaro treatment feels unclear, please get in touch with the Morrisons Clinic team. Our clinicians are available to support you through every step of your treatment journey.
You can contact us through your patient portal or via our support team directly. No question is too small, so please do get in touch.
Key takeaways
- Updated design: The pen has a longer plunger and new internal components to reduce leftover liquid after your fourth dose.
- Same medication: Your tirzepatide dose, strength and formulation have not changed.
- Gradual rollout: You may receive either the old or new pen design from April 2026. Both are safe and effective.
- No fifth dose: The new design makes it clear the pen is empty after your four weekly doses. Plan your next supply accordingly.
- Same routine: Your injection day, priming steps, injection sites and storage guidance are all unchanged.
References
How to Use the Mounjaro (tirzepatide) KwikPen, Eli Lilly and Company (2024). Accessed April 2026.
Eli Lilly KwikPen update announcement, March 2026.





